SoGAT Workshops
Meetings of the SoGAT group are typically held once a year. Participation is encouraged from blood and clinical laboratories, manufacturers of diagnostic assays and quality control reagents, providers of external quality assessment schemes, and regulatory and public health authorities. For more information or to sign up to our mailing list please contact us at sogat@nibsc.org
Update on SoGAT activities for 2020:
Thank you for your continuing interest in the activities of SoGAT. We know both from the levels of attendance and the feedback received from attendees at our meeting held in June 2019, that many people remain interested not only to hear about the work of this group, but also to actively contribute to the data and discussion that shape our actions.
At the same time, we recognise that there is a need to further increase awareness of our activities amongst scientists working in the area of the accurate measurement of infectious disease detection and burden.
As a result, when the SoGAT Scientific Advisory Group met in September 2019, we decided that that there will be no formal SoGAT workshop in 2020. Instead we will use the time and money saved to get out into the community and talk about SoGAT activities at other meetings and conferences where clinical diagnosis of infectious of infectious diseases is discussed. In the light of the COVID-19 pandemic, this decision may appear prescient
If you would like to assist in helping to spread the word about SoGAT then please contact us at sogat@nibsc.org. In addition, if you believe that there is an issue in infectious disease molecular diagnostics that would benefit from consideration by SoGAT, then please let us know.
The next SoGAT workshop will take place in 2021, with further details to be announced as soon as possible. In the meantime, we look forward to meeting colleagues who attend SoGAT at one or more of the following meetings this year, either in person or virtually:
| Meeting | Location | Date |
|---|
| WHO ECBS |
Geneva, Switzerland |
Oct |
| QCMD International Advisory Board meeting |
Glasgow, UK |
Oct |
| AMP 2020 Annual Meeting & Expo |
Vancouver, Canada |
17 - 21 Nov |
SoGAT 2019:
A two-day meeting was held at the MHRA headquarters in London, UK,on 3-4 June 2019.
Highlights included:
- Challenges for standardisation - commutability, extraction efficiency and genetic variability
- Using ddPCR and Next Generation Sequencing in diagnostics and standards development
- Point of impact testing - how can we standardise and the role of SoGAT
The meeting was attended by approximately 100 delegates working in infectious disease diagnostics.
WHO reference materials for NAT
World Health Organisation (WHO) biological reference materials provide a common set of standards which are used to ensure the quality of biological medicines worldwide.
International standards are manufactured according to WHO guidelines and are considered to be the ‘gold standard’ against which regional, national and international laboratories and manufacturers calibrate their own working standards.
They are calibrated in units of biological activity (International Units, IU) which are assigned following extensive studies involving multiple international laboratories. They are then formally established following review by the WHO Expert Committee on Biological Standardisation (ECBS) at its annual meeting in October.
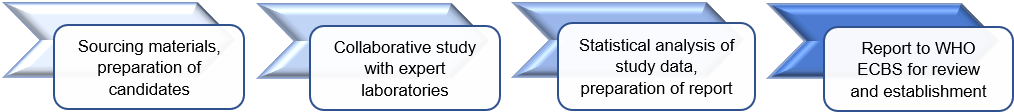
Recently established WHO reference materials for NAT for infectious diseases:
| WHO reference material | Product code | Date of establishment | WHO Collaborating Centre |
|---|
| Hepatitis C virus RNA for nucleic acid amplification techniques (6th WHO International Standard) |
18/184 |
October 2019 |
NIBSC |
| Human papilloma virus DNA types 6, 11, 31, 33, 45, 52,58 (1st WHO International Standards) |
14/256 (6),
14/100 (11),
14/258 (31),
14/260 (33),
14/104 (45),
14/262 (52),
14/264 (58) |
October 2019 |
NIBSC |
| HIV-1 p24 Antigen (1st WHO International Reference Panel) |
16/210 |
October 2018 |
NIBSC |
| HIV-2 p26 Antigen (VLP)-HIV-2 A7312 (1st WHO International Reference Reagent) |
16/236 |
October 2018 |
NIBSC |
| Human Immunodeficiency Virus type 2 RNA (HIV-2) for Nucleic Acid Amplification Techniques (2nd WHO International Standard) |
16/296 |
October 2018 |
NIBSC |
| Human Adenovirus DNA for nucleic acid amplification techniques (1st WHO international Standard) |
16/324 |
October 2018 |
NIBSC |
Further information on these NAT standards and others is available on the WHO website.
WHO International Standards for NAT for infectious diseases in development:
| WHO reference material | WHO Collaborating Centre | Status |
|---|
Aspergillus (1st WHO International Standard)
|
NIBSC |
Seeking offers of candidate materials
|
| Babesia (1st WHO International Standard) |
CBER |
- |
Enterovirus (1st WHO International Standard)
|
NIBSC |
Seeking offers of candidate materials
|
HIV, cell-associated (1st WHO International Standard)
|
NIBSC |
Seeking offers of candidate materials
|
HIV-1 CRF (2nd WHO International Reference Panel)
|
CBER/NIBSC |
Preparation of candidates |
| HSV-1/2 DNA (1st WHO International Standard) |
NIBSC |
Collaborative study completed.
Further commutability assessment underway |
| Influenza A RNA (1st WHO International Standard) |
NIBSC |
Pilot study scheduled |
| Influenza B RNA (1st WHO International Standard) |
NIBSC |
Pilot study scheduled
|
Leishmania (1st WHO International Standard)
|
NIBSC |
Seeking offers of candidate materials
|
Mycobacterium tuberculosis (1st WHO International Standard/panel)
|
NIBSC |
Seeking participants for collaborative study |
Plasmodium vivax (1st WHO International Standard)
|
NIBSC |
Seeking offers of candidate materials |
| RSV RNA (1st WHO International Standard) |
NIBSC |
Pilot study scheduled |
Trypanosoma cruzi (1st WHO International Standard)
|
NIBSC |
Seeking offers of candidate materials |
| VZV DNA (1st WHO International Standard) |
NIBSC |
Candidates scheduled |
WNV RNA (1st WHO International Standard)
|
NIBSC
|
Candidates being assessed in collaborative study |
History of SoGAT
Blood virology:
In 1994, the European Plasma Fractionation Association (EPFA), in conjunction with NIBSC, held a workshop to discuss the potential applications of NAT; from the detection of blood borne viruses in blood donation screening, to the production of “clean” plasma derived products. The meeting concluded that there was a need for standardization of NAT in the blood safety field, and that this warranted the formation of a group dedicated to standardization of NAT-based assays for blood borne viruses.
The 1st meeting of the International Working Group on the Standardisation of Genomic Amplification Techniques (SoGAT) for the virological safety testing of Plasma and Blood derived Products took place in April 1995. The group met to discuss standardisation techniques for blood viruses such as HCV, HBV, B19 as well as HIV. The rapid development of commercial assays for NAT and the appreciation of the scope of this new technology by many diagnostic and research laboratories, led to a decision that there was a need for reference materials for blood borne viruses.
Since 1994 the group have been paramount in the development of International Standards, reference panels and working reagents for HIV-1 RNA, HAV RNA, HCV RNA, HBV DNA and B19V DNA. Today, the group continues to support the development of new and replacement standards and reference panels and exchange information relevant to the scientific, technical and regulatory aspects of NAT.
Clinical diagnostics:
Using the experience and knowledge gained by the SoGAT group focusing on blood virology, a new SoGAT group was established to address the area of clinical virology, which remains largely unstandardised. The International SoGAT Clinical Diagnostics Working Group is focused on the standardisation of NAT assays used in the diagnosis of clinical pathogens. The group was established in parallel to the SoGAT Blood Safety Working Group in 2008 and the first meeting took place at NIBSC in June of that year. The group has supported the development of International Standards for HCMV, EBV, BKV, JCV, AdV and HHV-6 viruses for NAT, and new projects for HSV1/2, influenza A and B, RSV and enterovirus NAT standardisation.
Since 2013, the SoGAT blood and clinical groups have met at an annual joint workshop. Participation is encouraged from blood and clinical laboratories, manufacturers of diagnostic assays and quality control reagents, providers of external quality assessment schemes, and regulatory and public health authorities.
SoGAT Aims:
- Lead the development of WHO Reference Reagents and International Standards suitable for NAT and serological infectious disease assays (for screening of blood donations, plasma pool testing and diagnostics)
- Provide guidance on the preparation of external control materials calibrated against the WHO International Standards to be included in each run to ensure the reliability of the results
- Understand the relationship between clinical samples and the WHO ISs
- Promote standardisation of NAT and serological assays through inter-laboratory comparison studies or collaborations with EQA providers
- Provide a forum for the exchange of information to develop standards to support new technologies
- Provide a forum to react quickly to the standardisation needs of emerging or re-emerging pathogens.
Publications
The following articles highlight the work and successes of SoGAT or provide further information on the standardisation of NAT assays:
SoGAT - 25 years of improving the measurement of nucleic acids in infectious disease diagnostics (a review). Morris C, Govind S, Fryer J and Almond N. Metrologia. 2019 doi:10.1088/1681-7575/ab2aa3
WHO manual for the preparation of secondary reference materials for in vitro diagnostic assays designed for infectious disease nucleic acid or antigen detection: calibration to WHO International Standards. WHO Technical Report Series 2017. Geneva, Switzerland:WHO 2017; 1004, 389-455.
Standardisation of nucleic acid amplification assays used in clinical diagnostics: a report of the first meeting of the SoGAT Clinical Diagnostics Working Group. Fryer JF, Minor PD. J Clin Virol. 2009 Feb;44(2):103-5. doi: 10.1016/j.jcv.2008.11.007.
Stay up to date
For more information or to sign up to our mailing list please contact us at sogat@nibsc.org.